Proteins: Essential Building Blocks of Life.pptx
- 1. Protein
- 2. Introduction • Most important of cell constituent. • It is made up of amino acids. • It is responsible for almost every function that occurs in the body • A regular and adequate supply of protein in a diet is essential for cell integrity and function. • Dietary proteins are the primary source of nitrogen that is metabolized by the body. • Adult = 70 to 100 gram per day • Amino acids are the structural unit of proteins which have carboxyl group (-COOH) an amino acid group (-NH2) bound to the same carbon atom called α-carbon. • Amino acids differ from each other in their side chains or R- groups attached to the carbon atoms. • Amino acids join by peptide bonds to form proteins. • There are 20 amino acids involve in formation of proteins. • Theses 20 amino acids are referred as standard amino acids.
- 3. Classification • Essential amino acids • Non essential amino acids Nutritional requirement of amino acids: • Glucogenic amino acids • Ketogenic amino acids • Both glucogenic and ketogenic Metabolic fate of amino acids:
- 4. Essential amino acids • Most microorganisms can synthesize 20 amino acids whereas human cannot synthesize 10 of them because some of the enzymes required for the synthesis of this amino acids are not present in the human body. • This 10 amino acids must be supplied through the diet, which are known as Essential amino acids • Ten amino acids, essential for humans include: • Among the 10 amino acids, arginine and histidine are known as semi essential amino acids since this amino acids are synthesized partially in human body. • It is essential in diet during periods of rapid growth as in childhood and pregnancy. • Deficiency of one amino acids impairs protein synthesis and lead to negative nitrogen balance. Phenylalanine Valine Threonine Tryptophan Isoleucine Methionine Lysine Leucine Histidine Arginine
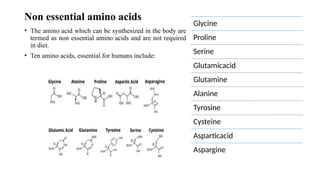
- 6. Non essential amino acids • The amino acid which can be synthesized in the body are termed as non essential amino acids and are not required in diet. • Ten amino acids, essential for humans include: Glycine Proline Serine Glutamicacid Glutamine Alanine Tyrosine Cysteine Asparticacid Aspargine
- 7. Metabolic fate of amino acids: 1. Glucogenic amino acids • Which can be converted into glucose • 14 out of 20 amino acids are glucogenic amino acids, some of them are glycine, alanine, serine, valine, cysteine, methionine, threonine, etc. 2. Ketogenic amino acids • Which are converted to ketone bodies • Two amino acids leucine and lysine are ketogenic. 3. Both glucogenic and ketogenic • Which are converted to both glucose and ketone bodies • Four amino acids isoleucine, phenylalanine, tryptophan and tyrosine are glucogenic and ketogenic.
- 8. Digestion and Absorption of Proteins • Proteins are large size molecules. • They are polymers of amino acids. • Dietary proteins are very large complex molecules. • That cannot be absorbed from the intestine. • To be absorbed, dietary proteins must be digested to small simple molecules, which are easily absorbed from the intestine. • There is no digestion of protein in mouth, it starts in stomach.
- 9. Digestion in stomach When protein enters the stomach, it stimulates the secretion of gastrin hormone, from gastric mucus cells, which in turn, stimulates the release of gastric juice containing hydrochloric acid, pepsinogen and rennin in infants. 1. Hydrochloric acid: • It provides an acid environment for the action of pepsin. 2. Pepsin • It is secreted in an inactive form, pepsinogen. • It is converted into active pepsin in the gastric juice by the enzymatic action of pepsin itself or by high hydrogen ion concentration. • Pepsin cleaves long polypeptide chain into a mixture of smaller peptides and some free amino acids. 3. Rennin (infants) • It is important in the digestive process of infants. • It is absent in adults rennin is also called chymosin and rennet. • Action of rennin is to clot milk. The purpose of this action is to convert milk into a more solid form to prevent the rapid passage of milk from the stomach of infants.
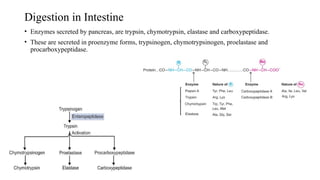
- 10. Digestion in Intestine • Enzymes secreted by pancreas, are trypsin, chymotrypsin, elastase and carboxypeptidase. • These are secreted in proenzyme forms, trypsinogen, chymotrypsinogen, proelastase and procarboxypeptidase.
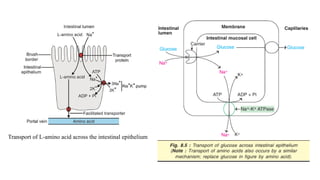
- 11. Absorption of amino acids • The absorption of most amino acids involves an active transport mechanism requiring ATP and specific transport protein in the intestinal mucosal cells. • Many transporters have Na+ dependent mechanisms, coupled with NA+ K+ pumps, similar to those describe for glucose absorption. • Amino acids, release by digestion, pass from the gut through hepatic portal vein through liver
- 12. Transport of L-amino acid across the intestinal epithelium
- 13. Metabolism of Proteins and Related Disorders • Protein is digested an broken down to amino acids which are absorbed into circulation and taken to the liver. • Protein metabolism means various biochemical processes responsible for the synthesis and breakdown of amino acids. • Primary role – serve as building blocks of synthesis of tissue protein and other biologically important compounds. • Amino acids in excess cannot be stored in contrast with fatty acid and glucose, nor are they excreted. • The liver is the primary site of all amino acid catabolism. • The synthesis of plasma proteins albumin, fibrinogen and globulin also occurs in liver.
- 14. Metabolism of Amino Acids • In the catabolism amino acids, amino acid- looses amino group in the form of ammonia – excreted – urea in the liver by the reaction of urea cycle. • Remaining carbons skeleton, α- Ketoacids – undergo oxidation to CO2 & H2O by the reaction of citric acid cycle.
- 15. Catabolism of amino acids Complete catabolism of amino acids includes 3 stages: 1. Removal of α- amino group in a form of ammonia by: a. Transamination by the enzyme amino transferase also called transaminase. • Transfer of an amino ( NH2) group from an amino acid to a keto acid b. Deamination by glutamate dehydrogenase. • Removal of amino group in a form of ammonia (Pyridoxal phosphate ) Deamination
- 16. … 2. Disposal of ammonia in a form of urea in the liver by reaction urea cycle. 3. Disposal of carbon skeleton of amino acids after removal of α- amino group. • The carbon skeleton of 20 amino acids are converted into compounds which are intermediate of TCA cycle. • This carbon skeletons are used for gluconeogenesis or ketogenesis or are completely oxidized to CO2 & H2O.
- 17. Fate of amino acid-carbon skeleton 1. Amino acids – degraded to acetyl CoA termed as ketogenic (leucine and lysine). 2. Amino acids – degraded to pyruvate, α- ketoglutarate, succinyl CoA, fumarate or oxaloacetate termed as glucogenic. 3. Isoleucine, phenylalanine, tryptophan and tyrosine are both glucogenic and ketogenic. 4. Other 14 amino acids are classed as purely glucogenic. Metabolic fate of carbon skeleton of amino acids
- 18. Urea Cycle • Ammonia is converted to urea by “Kreb’s Henseleit urea cycle” which is exclusively occurs in liver. • The first two reactions of urea cycle occurs in mitochondria, whereas remaining reactions occur in the cytosol. • The sequence of reactions involved in the biosynthesis of urea, summarized in five steps. 1. Formation of carbamoyl phosphate: 2. Formation of citrulline 3. Formation of argininosuccinate 4. Formation of arginine and fumarate 5. Formation of urea and ornithine
- 19. Reaction of Urea cycle
- 20. Plasma Proteins • Plasma proteins are proteins present in blood plasma. • Plasma is the clear yellowish fluid portion of blood (55-60%) in which cell are suspended. • Plasma obtained by centrifuging blood that has been treated with anticoagulant to prevent clotting. • Plasma contains a variety of proteins with different functions. • Changes in the concentration of plasma proteins can give important clinical information.
- 21. Types of Plasma Proteins • There are major three different types of plasma proteins, each serving different specific functions. 1. Albumin: major protein in plasma and exclusively synthesized by liver 2. Globulins with subtypes o α-globulins o β-globulins o γ-globulins 3. Fibrinogens
- 22. Functions of Plasma Proteins • Maintenance of colloidal osmotic pressure • Maintenance of blood pressure • Maintenance of acid-base balance • Role in transport of metabolites • Role of protein in the defense mechanism of the body • Involvement in inflammatory responses • Enzymatic function of proteins • Role in blood coagulation
- 23. Normal Value of Plasma Proteins • The normal value of plasma proteins are: o Total protein: 6 to 8g o Albumin (A): 3.5 to 6g o Globulin (G): 2 to 3.5g oFibrinogen: 200 to 400mg oA/G ratio: 1:2:1 to 2:5:1
- 24. Proteinuria: Hypoproteinemia, Hyperglobulinemia • Normally, the total amount of protein excreted in urine over 24 hours is less than 150mg and is not detectable by routine tests. • Presence of protein in urine in excess is referred as proteinuria. • An excess protein in urine is suggestive of illness. • Protein excreted is mostly albumin and some smaller proteins. • With severe proteinuria, general hypoproteinemia can develop which results in diminished oncotic pressure. • Diminished oncotic pressure leads to edema.
- 25. Causes of Proteinuria Proteinuria can caused by: increased permeability of glomerular membrane Reduced tubular reabsorption Increased concentration of low molecular weight proteins in the plasma Abnormal secretion of protein from the renal tract.
- 26. Types of Proteinuria 1. Glomerular proteinuria: Most common and serious type of proteinuria because most of excreted protein is albumin glomerular proteinuria often referred as albuminuria. Protein in urine is the first sign of glomerular injury. Increased filtration of macromolecules (such as albumin) across the glomerular capillary wall. Diabetic nephropathy, glomerulopathy, exercise induced, orthostatic proteinuria Excretion of albumin in the range of 30 to 300mg/day is termed as micro-albuminuria. Micro-albuminuria is the earliest sign of renal damage due to diabetes mellitus and hypertension. In normal pregnancy, urine protein excretion may increase slightly, more severe proteinuria occurs in pre-eclamptic toxemia. Functional or benign proteinuria is a form of glomerular proteinuria that is probably due to changes of blood flow through the glomeruli. It is seen with exercise, fever, heart failure, hypertension, etc.
- 27. 2. Tubular proteinuria: It is characterized by appearance of low molecular weight proteins in the urine due to reduced tubular reabsorption. A low molecular weight protein typically excreted in tubular proteinuria include; β-macroglobulin, retinol binding protein lysosome, etc. Tubular proteinuria commonly associated with glomerular proteinuria. When tubular proteinuria occurs alone, albumin excretion is only slightly increased. Acute tubular proteinuria may occur with burns and chronic tubular proteinuria occurs in cirrhosis. 3. Overload proteinuria: Overload proteinuria is due to increased concentration of abnormal low molecular weight proteins in the plasma, such as immunoglobulin light chain. For example, high plasma concentration of immunoglobulin light chain is seen in multiple myeloma.
- 28. 4. Post-renal proteinuria: It is due to abnormal secretion of protein into urinary tract. Post-renal proteinuria refers to protein arising from the urinary tract below the kidneys and usually due to inflammation or cancer. Lesions of urethra, renal pelvis, bladder and prostate can cause such proteinuria.
- 29. Hypoproteinemia • It is a condition where there is an abnormally low level of protein in blood. • Decreased level of plasma albumin are seen in various disorders. • Decreased serum protein reduces the osmotic pressure of blood, leading to loss of fluid from intravascular compartment, or blood vessels, to intestinal tissues, resulting in edema Causes: • Not enough protein in diet • You can become deficient in protein if you don’t eat enough food sources. Severe protein deficiency is called kwashiorkor, a type of energy malnutrition affecting young children. • Malabsorption of proteins • Liver disease can also cause hypoproteinemia by decreasing the synthesis of plasma proteins like albumin. • Renal diseases like nephrotic syndrome can also result in hypoproteinemia because substances like protein, which need to stay in your blood, leak into your urine. An excess of protein in your urine due to kidney damage is called proteinuria. • Plasma proteins are lost in urine. Whole body infection (sepsis) resulting in hypoproteinemia.
- 30. Hyperglobulinemia • It is an abnormal condition characterized by increased levels of certain immunoglobulin in the blood. • Immunoglobulins are antibodies in blood and tissues that work to remove bacteria, viruses, fungi and foreign substances from the blood. • There are different types of antibodies in your blood, IgG, IgA, IgM, IgD, and IgE. The most common antibody is Immunoglobulin G (IgG). People with hypergammaglobulinemia most often have increased levels of IgG. • Multiple myeloma, a plasma cell cancer results due to abnormally high concentration of serum immunoglobulins, usually IgG or IgA. • In multiple myeloma, light chains are produced in excess than heavy chains enter the blood stream, because they are relatively low molecular weight, they pass through glomerular membrane and appear in urine. • Protein chains of low molecular weight are known as Bence-Jones proteins. • Bence-Jones proteins have remarkable characteristic of precipitating on heating urine from 45° to 60°C and redissolved heating is continued above 80°C. • Multiple myeloma with Bence-Jones protein in urine is called “Light chain disease.”
- 31. Electrophoresis • The movement of charged particles through an electrolyte when subjected to an electric field. • This method is widely used for the separation of proteins in biological fluids. • Factors Affecting Electrophoresis • Net charge on the particles • Mass and shape of the particles • The pH of the medium • Strength of electrical field • Properties of the supporting medium • Temperature
- 32. Components of electrophoresis • Buffer-barbitone buffer (8.6) • At pH 8.6 serum proteins will have net negative charge and will migrate towards the anode. • Support medium • Filter paper • Cellulose acetate membrane • Agar • Polyacrylamide gel electrophoresis • Visualization of protein bands • Fixation (acetone, methanol) • Dye (naphthalene black, Coomassie blue)
- 33. Electrophoresis of normal serum sample • Albumin • Alpha 1 • Alpha 2 • Beta • Gamma
- 36. In born errors of amino acid metabolism
- 37. …
- 38. …
- 39. …
- 40. …